Taxation Ruling
TR 93/40A - Addendum
Income tax: imputation of company tax: form approved by Commissioner of Taxation for issue to shareholders receiving dividends for 1994-95 and later franking years
-
Please note that the PDF version is the authorised version of this ruling.View the consolidated version for this notice.
FOI status:
May be releasedAddendum
1. This Addendum provides additional guidelines on the approved form of dividend statements that an Australian resident company must provide to shareholders when paying a dividend.
2. These guidelines apply when a company pays a dividend that consists, either wholly or partly, of a 'foreign dividend account declaration amount' ["FDA amount"]. An FDA amount results from the declaration, in accordance with section 128TC, of a 'foreign dividend account declaration percentage' ["FDA percentage"] by the company paying the dividend.
3. A company can declare an FDA percentage if it has derived certain foreign-source dividend income. The effect of the declaration is to exempt a non-resident recipient of the dividend from dividend withholding tax to the extent the dividend consists of an FDA amount. A non-resident is also exempt from withholding tax to the extent the dividend also consists of a franked amount.
Changes to Taxation Ruling TR 93/40 Ruling
4. The reference to 'the attachment' in paragraph 3 is to be read as referring to the appropriate attachment. This addendum inserts an additional attachment "Attachment B" which sets the approved form of statements if the dividend consists wholly or partly of an FDA amount.
Decimal places on shareholder dividend statement
5. The following sentence is added to the end of paragraph 5 of the Ruling:
"If there is an FDA amount less than the entire amount of the dividend, it may also be rounded off to the nearest cent."
Explanations
6. The following paragraphs are added after paragraph 9:
"Dividends that include an FDA amount
9.1 From 1 July 1994, a company that has a surplus in its foreign dividend account may pay a dividend that consists of an FDA amount. If the dividend is an unfranked dividend, it may consist wholly or partly of an FDA amount. If the dividend is franked, the FDA amount is limited to the amount of the dividend that has not been franked.
9.2 For a dividend that consists entirely of an FDA amount, a company must specify (in addition to any of the information mentioned at paragraphs 8 and 9 of the Ruling) that the dividend is exempt from Australian withholding tax if it is derived by a non-resident [paragraph 128TD(1)(a)]. See Forms 4 and 7, Attachment B for an approved form of statement.
9.3 For a dividend that consists partly of an FDA amount, paragraph 128TD(1)(b) requires a company to specify (in addition to the information mentioned at paragraphs 8 and 9 of the Ruling):
- (a)
- the amount of the dividend that is exempt from Australian withholding tax if it is derived by a non-resident;
- (b)
- the amount, if any, of the dividend that is not exempt from Australian withholding tax if it is derived by a non-resident;
- (c)
- the amount, if any, of withholding tax deducted from the dividend. For the requirements in relation to an unfranked dividend see Forms 5 and 8, Attachment B for an approved form of statement. For the requirements in relation to a partly franked dividend, see Forms 6 and 9, Attachment B for an approved form of statement. As Forms 5 and 6 show, this information may be incorporated in statement in the approved form required by section 160AQH.
9.4 If the information is not incorporated in a statement in the approved form required by section 160AQH, the statement containing the information required by section 128TD must also contain the following additional information:
- (a)
- the name of the company paying the dividend;
- (b)
- the date of payment of the dividend; and
- (c)
- the name of the shareholder."
Attachment A
7. The following sentence replaces the introductory sentence of Attachment A:
"The following are approved formats for the statements to be completed by Australian resident companies that pay dividends to shareholders, other than dividends that consist of FDA amounts."
Attachment B
8. Taxation Ruling TR 93/40 is supplemented by an additional attachment 'Attachment B' as set out below.
Attachment B
The following are approved formats for the statements to be provided by Australian resident companies that pay dividends to shareholders where the dividends consist of FDA amounts.
Form 4 - For unfranked dividends consisting entirely of an FDA amount, only
This form satisfies both section 160AQH and section 128TD requirements.

Form 5 - For unfranked dividends consisting partly of an FDA amount, only
This form satisfies both section 160AQH and section 128TD requirements.

Form 6 - For partially franked dividends or for general use
This form satisfies both section 160AQH and section 128TD requirements.
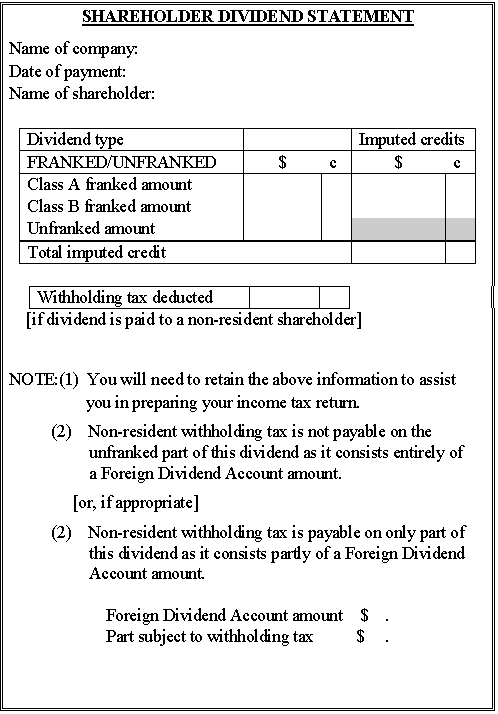
Form 7 - For unfranked dividends consisting entirely of an FDA amount, only
This form may be used where the statement given to satisfy section 128TD requirements is separate from that given to satisfy section 160AQH requirements.

Form 8 - For unfranked dividends consisting partly of an FDA amount, only
This form may be used where the statement given to satisfy section 128TD requirements is separate from that given to satisfy section 160AQH requirements.

Form 9 - For partially franked dividends or for general use
This form may be used where the statement given to satisfy section 128TD requirements is separate from that given to satisfy section 160AQH requirements.
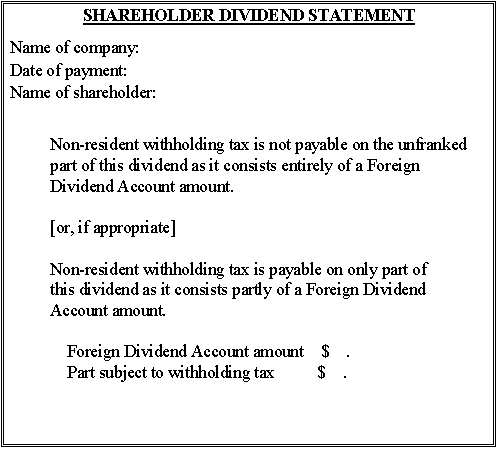
Form 10 - Additional statement for dividends paid during the transitional period from 1 July 1994 to the date of enactment of the amending legislation
This statement must accompany any dividend paid before the date of commencement of the Taxation Laws Amendment Act (No. 3) 1994 if it is to consist of an FDA amount.
It may either be given to shareholders as a separate statement or incorporated in shareholder dividend statements using one of Forms 4 to 9.

Date of effect
This Addendum applies to dividends paid by resident companies, after 1 July 1994, that consist partly or wholly of an FDA amount.
Commissioner of Taxation
7 July 1994
